 This partial charge separation makes SO2 a polar molecule. Here the electron distribution is unevenly distributed between the sulfur and oxygen atoms.Īs a result, the oxygen atoms have a partial negative charge, while the sulfur atom has a partial positive charge. The polar covalent bonds and the bent geometry of SO2 create a dipole moment. In the SO2 Lewis Structure, the two oxygen atoms have two lone pairs of electrons, causing them to repel each other and create a bent molecular geometry. The electronegativity difference between sulfur and oxygen is 1.5, indicating a polar covalent bond. The SO2 molecule is polar due to its asymmetrical shape and polar covalent bonds. Explanation of the polar nature of SO2 molecule The total number of valence electrons in the SO2 Lewis Structure is 18. Each oxygen atom should have two lone pairs of electrons above and below the plane of the molecule. To represent the SO2 Lewis Structure, draw the sulfur atom in the center, with two lines connecting it to the oxygen atoms on either side. Each oxygen atom has two lone pairs of electrons. The sulfur atom is in the center with two oxygen atoms attached by a double bond. The SO2 Lewis Structure consists of one sulfur atom and two oxygen atoms, with a double bond between sulfur and each oxygen atom. Representation of the SO2 Lewis Structure Count the total number of electrons in the Lewis structure to ensure it matches the total number of valence electrons.Ĭ. 
If not, move lone pairs from outer atoms to form double or triple bonds until all atoms have a full octet. Check that all atoms have a full octet (except hydrogen, which only needs two electrons).Place the remaining electrons as lone pairs on the outer atoms first, then on the central atom.Subtract the electrons used in step 3 from the total number of valence electrons to determine the number of electrons remaining.Attach the surrounding atoms to the central atom by single bonds.Identify the central atom, which is usually the atom with the lowest electronegativity.Determine the total number of valence electrons present in the molecule by adding up the valence electrons of all the atoms.Steps in drawing the SO2 Lewis Structureĭrawing the SO2 Lewis Structure involves several steps: 
The SO2 Lewis Structure also helps in predicting the molecule’s geometry, which is bent due to the presence of two lone pairs on the oxygen atoms. The double bond between sulfur and oxygen represents the sharing of two electrons, while the lone pairs on each oxygen atom represent the non-bonding electrons. The SO2 Lewis Structure helps in understanding the bonding and non-bonding electrons present in the molecule. Therefore, the total number of valence electrons in SO2 is 18. The sulfur atom has six valence electrons, while each oxygen atom has six valence electrons. In the SO2 Lewis Structure, the sulfur atom is placed in the center, and the two oxygen atoms are attached to it by a double bond. 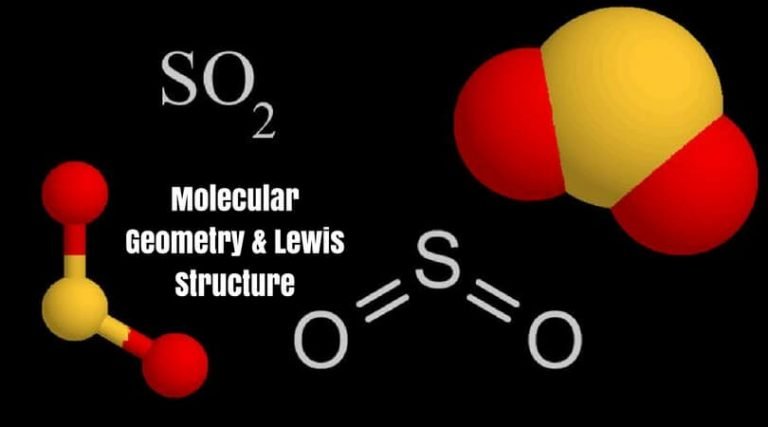
To draw the SO2 Lewis Structure, the valence electrons of the sulfur and oxygen atoms are considered, and the electrons are arranged to minimize their repulsion. The SO2 Lewis Structure is a graphical representation of the molecule’s electron distribution. Sulfur dioxide is a colorless gas with a pungent odor and is commonly used in the production of sulfuric acid. When sulfur burns in the air, it reacts with oxygen to produce sulfur dioxide. It consists of one sulfur atom and two oxygen atoms. The chemical formula of sulfur dioxide is SO2. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
